|
Lead
Exposure & Alzheimer’s Disease: Is There A Link?
A fact sheet for medical professionals
by
Dr. Iman Hegazi, MBBS, MD. Forensic Medicine & Toxicology,
Global Lead Advice & Support Service (GLASS), run by The
LEAD Group Inc
Alzheimer's
disease (AD), or simply Alzheimer's, is the most common form of
dementia. This incurable, degenerative, and terminal disease was
first described by German psychiatrist and neuropathologist
Alois Alzheimer in 1906 and was named after him. (1) Generally
it is diagnosed in people over 65 years of age, although the
less-prevalent early-onset Alzheimer's can occur much earlier.
An estimated 26.6 million people worldwide had Alzheimer's in
2006; this number may quadruple by 2050. (2,
3)
The
term "Alzheimer’s" is rapidly being used by the
general public to refer to any elderly person suffering from
dementia. Everyone who suffers from Alzheimer's disease has
dementia. However, not everyone with dementia has Alzheimer's
disease. Dementia may be caused by several other diseases.
History:
Alzheimer’s
disease, the chronic decline in intellectual capabilities, was
recognized by the ancient Greek physician Hippocrates. At the
time "senility" was thought to be simply a normal part
of aging (4). Alois Alzheimer, better defined the disease in a
speech given in November 1906. Dr. Alzheimer examined the brain,
after her death, of a woman who died at the age of 56, after
having paranoid delusions, hallucinations, and a loss of memory,
and noticed senile plaques and neurofibrillary tangles in the
neurons (brain cells) (5). Alzheimer wrote, "On the whole,
it is evident that we are dealing with a peculiar, little known
disease process." Up until 1970 the diagnosis of
Alzheimer’s disease was restricted to patients less than 65
years old(4). Alzheimer’s disease is no longer defined by age
with between 50% and 75% of all dementia cases now being
diagnosed as Alzheimer’s disease, with the proportion
increasing with age (6,7).
Overview
of Changes in the Brain:
Alois
Alzheimer was able to describe two of the major changes in brain
structure caused by Alzheimer’s disease. In addition to the
neurofibrillary tangles and neuritic plagues, AD also results in
granulovascular degeneration, brain shrinkage, and decreased
amounts of neurotransmitters (brain chemicals involved in
communication). All of these changes impair the function of
brain cells (neurons) and eventually lead to cellular death.
Unlike many other cells, the brain is unable to regenerate new
neurons. The gradual and progressive death of neurons is
mirrored in specific behaviour changes. (4)
Phases
of Alzheimer's
Mild
dementia
The
chief characteristic of mild Alzheimer’s disease is
forgetfulness. Not only will the individual misplace items but
will start to place them in unusual locations (e.g. keys in the
freezer). The decline may be so gradual that it may take family
members years to notice since some forgetfulness is a natural
part of aging. Loss of a sense of time and direction greatly
interferes with the ability to navigate and are important
components of a "sense of direction." The
characteristic of becoming lost, especially in unfamiliar
territory is a common hallmark in mild AD. (4)
Moderate
dementia
Once
Alzheimer’s disease deteriorates to the moderate stage;
profound memory loss that interferes with daily activities
characterizes the disorder. The AD patient in the moderate stage
is dependent upon others. The sufferer becomes lost even in
familiar surroundings and has lost the ability to learn any new
material. (4)
Severe
dementia
Once
the disease progresses to the severe stage the patient suffers
from severe impairment of mind (cognitive functions) and body.
The subject has reached the point they may no longer recognize
their spouse and children. The AD patient no longer has any
sense of time or current location. Verbal communication has
decreased to the point of phrases, words, or merely syllables
are constantly repeated. Eventually, even this simple
communication degenerates to a complete inability to speak. The
overall loss of coordination and body control means the patient
may require help dressing, bathing, grooming, eating, and
toileting. (4)
|
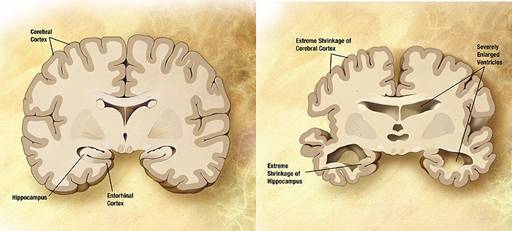
Description:
Combination of two diagrams of the brain in one for
comparison. Left: A normal brain. Right: The brain of a
person with Alzheimer's disease showing extreme
shrinkage of the cerebral cortex and hippocampus and
enlargement of the ventricles. (8)
The
copyright holder of this work grant anyone the right to
use this work for any purpose , without any conditions,
unless such conditions are required by law. http://en.wikipedia.org/wiki/File:COMPARISONSLICE_HIGH.JPG |
Although
Alzheimer’s disease (AD) is a progressive neurodegenerative
disorder whose clinical manifestations appear in old age, the
initial events that trigger this disease may begin very early in
life.
In
consequence of the “Barker hypothesis”, which links early
life experiences and adult diseases, there is a new concept
regarding certain adult diseases that emphasizes the role of
environmental factors operating during the preconceptual, fetal,
and infantile phases of life. (9)
The
sporadic nature of most AD cases strongly argues for an
environmental link that may accelerate normal age-related
processes in the brain. Due to the widespread occurrence of AD,
such environmental agents (risk factors) would have to be
widespread and persistent, and chronic human exposure to them
very common. Although few environmental agents may fit this
description, the heavy metal Pb is widespread and is known to
produce permanent behavioral and cognitive perturbations. (10,
11)
Lead
(Pb) poses widespread public concern. Humans can be exposed to
Pb through paint, glazed earthenware, lead piping, solder in
food containers, moonshine whiskey, and automobile battery
casings. Occupational exposure to Pb can come from smelters,
battery manufacturing, welding, automobile radiator repair, and
production of Pb-based paints. Although organic forms of Pb have
been removed from gasoline, inorganic Pb still remains the
number one environmental hazard facing humans today. (12)
Occupational
lead exposure may have long-term effects and dramatically
increase the risk of developing Alzheimer's disease in later
years, according to research presented during the American
Academy of Neurology's 52nd Annual Meeting in San Diego, CA,
April 29 -- May 6, 2000. Researchers believe that people who
have worked in jobs with high levels of lead exposure are up to
3.4 times more likely to develop Alzheimer's disease.
Dr Koss of the American Academy of Neurology quotes:
"Although lead has long been known to be toxic -- and is
believed to have affected the brains of some of the rulers of
the Roman Empire, thereby causing its downfall -- its long-term
damages are difficult to measure, and thus, the extent of its
negative effects have been largely overlooked." (13)
The
pathogenesis of Alzheimer’s disease: Evidence for an
environmental link:
AD
pathology is characterized by senile plaques and neurofibrillary
tangles (NFTs), combined with massive neuronal loss. The major
constituents of senile plaques (or amyloid plaques) are 39–42
residue peptides (Aβ) snipped from a larger protein called
the amyloid precursor protein (APP). Current studies indicate
that APP is processed by a group of secretases. Cleavage of APP by β-secretase produces sAPPβ
and C99 fragments. C99 is then cleaved by γ-secretase to
release Aβ. However, prior processing of APP by α-secretase
precludes the formation of the neurotoxic Aβ. Inhibiting
α- secretase activity predisposes the processing of APP via
the β-secretase pathway, which ultimately leads to
neurotoxicity. On the other hand, suppression of β-secretase
or γ-secretase activity prevents the formation of the
neurotoxic Aβ molecule. (14,15)
Aβ
processing and aggregation are thought to be critical events in
the etiology of neurodegenerative diseases such as AD. Mutations
in APP have been shown to promote AD pathogenesis in familial AD
by altering its proteolytic processing, which results in an
increased production of Aβ. (16)
Recent
studies in rodents have shown that exposure to lead (Pb) during
brain development predetermined the expression and regulation of
the amyloid precursor protein (APP) and its amyloidogenic β-amyloid
(Aβ) product in old age. It is also reported that the
expression of AD-related genes [APP, BACE1] as well as their
transcriptional regulator (Sp1) were elevated in 23-year-old
monkeys exposed to Pb as infants leading to an Alzheimer's
disease (AD)-like pathology in the aged monkeys. (17, 18)
Chronic
lead (Pb) exposure also affected granule cell morphology in Pb-exposed rats. Dendrites frequently appeared dystrophic,
similar to those present in Alzheimer’s disease. Dendrites
play an essential role in neuronal signaling and aberrations in
dendritic morphology are likely to alter their functional
characteristics. (19)
These
data suggest that AD pathogenesis is influenced by early life
exposures and argue for both an environmental trigger and a
developmental origin of AD. The findings implicate an
environmental agent (Pb) in the pathogenesis of AD and
demonstrate that development is an important period of
vulnerability, which could increase future susceptibility to
neurodegeneration and AD pathology.
Therapeutic
proof of heavy metal implication:
Clioquinol,
a chelating agent used in heavy metal toxicity, is under
investigation for the treatment of Alzheimer disease. A pilot
phase 2 study with clioquinol showed that the effect of the
treatment was significant in the more severely affected group
and supports further investigation of this novel treatment.
Clioquinol is a chelator that crosses the blood-brain
barrier and may diminish the accumulation of amyloid beta in
plaques. Clioquinol was shown to produce a significant reversal
of amyloid-beta plaque deposition in vitro as well as in vivo in
a clinical trial. (20, 21) Another pilot study has shown the
efficacy of the chelating agent d-penicillamine in reducing
oxidative stress in Alzheimer disease patients. (22)
In
conclusion, lead (Pb) is a xenobiotic metal with no known
essential function in cellular growth, proliferation, or
signalling and there is compelling evidence that exposures to Pb
have adverse effects on the nervous system. (23) The sporadic
nature of Alzheimer's disease (AD) and a number of studies
suggest that AD pathogenesis is influenced by early life
exposures and argue for both an environmental trigger and a
developmental origin of AD. Recent research presents novel
findings that implicate lead (Pb) in the pathogenesis of AD and
demonstrate that development is an important period of
vulnerability, which could increase future susceptibility to
neurodegeneration and AD pathology.
References
-
Berchtold NC, Cotman CW (1998). "Evolution in the
conceptualization of dementia and Alzheimer's disease:
Greco-Roman period to the 1960s". Neurobiol. Aging 19
(3): 173–89. doi:10.1016/S0197-4580(98)00052-9.
PMID
9661992.
-
Brookmeyer
R, Gray S, Kawas C (September 1998). "Projections
of Alzheimer's disease in the United States and the public
health impact of delaying disease onset". Am J
Public Health 88 (9): 1337–42. doi:10.2105/AJPH.88.9.1337.
PMID
9736873.
-
Brookmeyer
R, Johnson E, Ziegler-Graham K, MH Arrighi (July 2007).
"Forecasting
the global burden of Alzheimer’s disease".
Alzheimer's and Dementia 3 (3): 186–91. doi:10.1016/j.jalz.2007.04.381.
http://works.bepress.com/cgi/viewcontent.cgi?article=1022&context=rbrookmeyer.
-
Alzheimer's
Disease and Related Disorders SAR Research
Alzheimer's Overview www.dbs-sar.com/SAR_Research/alzheimer_research.htm
-
German
E Berrios (June 2004) “The history of 'Alzheimer's
disease'” The Human Genome23/6/2004 http://genome.wellcome.ac.uk/doc_WTD020951.html
-
American
Psychiatric Association (2006) Practice
guidelines for the Treatment Of Psyciatric Disorders
Compendium 2006
-
Alewijn
Ott, Monique M B Breteler, Frans van Harskamp, Jules J
Claus, Tischa J
M van der Cammen, Diederick
E Grobbee, Albert Hofman (1995) “Prevalence of Alzheimer's
disease and vascular dementia: association with
education.” The Rotterdam study BMJ 1995;310:970-973 (15
April) www.bmj.com/cgi/content/full/310/6985/970
-
ADEAR
(Alzheimer's Disease Education & referrals center)http://en.wikipedia.org/wiki/File:COMPARISONSLICE_HIGH.JPG
-
Barker
DJ, Winter PD, Osmond C, Margetts B, Simmonds SJ (1989)
Weight in infancy and death from ischaemic heart disease.
Lancet 2:577–580. www.thelancet.com/journals/lancet/article/PIIS0140-6736(89)90710-1/abstract
-
Yang,
Y., Ma, Y., Ni, L., Zhao, S., Li, L., Zhang, J., Fan, M.,
Liang, C., Cao, J., and Xu, L. (2003) Lead
exposure through gestation-only caused long-term
learning/memory deficits in young adult offspring. Exp.
Neurol. 184, 489–495.
-
Weisskopf,
M. G., Hu, H., Mulkern, R. V., White, R., Aro, A., Oliveira,
S., and Wright, R. O. (2004) Cognitive deficits and magnetic
resonance spectroscopy in adult monozygotic twins with lead
poisoning. Environ. Health Perspect. 112, 620–625.
www.pubmedcentral.nih.gov/picrender.fcgi?artid=1241931&blobtype=PDF
-
Goyer,
R. A. (1996) Toxic effects of metals, in Casarett and
Doull’s Toxicology: The Basic Science of Poisons. Casarett
and Doull’s Toxicology: The Basic Science of Poisons (Klaassan
C. D., ed) pp. 691–736, McGraw Hill, New York.
-
Gray
Environmental, Inc. Study on the connection between lead
exposure and Alzheimer’s disease. May 4, 2000. www.grayenvironmental.com/lead%20exposure%20and%20alzheimers.htm
-
Hussain,
I., Powell, D., Howlett, D. R., Tew, D. G., Meek, T. D.,
Chapman, C., Gloger, I. S., Murphy, K. E., Southan, C. D.,
Ryan, D. M., et al. (1999) Identification
of a novel aspartic protease (Asp 2) as beta-secretase.
Mol. Cell. Neurosci. 14, 419–427. www.ncbi.nlm.nih.gov/pubmed/10656250
-
Christensen,
M. A., Zhou, W., Qing, H., Lehman, A., Philipsen, S., and
Song, W. (2004) Transcriptional regulation of BACE1, the
beta-amyloid precursor protein beta-secretase, by Sp1. Mol.
Cell. Biol. 24, 865–874. www.pubmedcentral.nih.gov/picrender.fcgi?artid=343820&blobtype=PDF
-
Ross,
C. A., and Poirier, M. A. (2004) Protein aggregation and
neurodegenerative disease. Nat. Med. 10, S10–S17 www.nature.com/nm/journal/v10/n7s/PDF/nm1066.PDF
-
Basha,
M. R., Wei, W., Bakheet, S. A., Benitez, N., Siddiqi, H. K.,
Ge, Y., Lahiri, D. K., and Zawia, N. H. (2005) The fetal-basis of
amyloidogenesis: exposure to lead and latent
over-expression of amyloid precursor protein and β-amyloid
in the aging brain. J. Neurosci. 25, 823–829.
www.jneurosci.org/cgi/reprint/25/4/823
-
Wu
J, Basha
MR, Brock
B, Cox
DP, Cardozo-Pelaez
F, McPherson
CA, Harry
J, Rice
DC, Maloney
B, Chen
D, Lahiri
DK, Zawia
NH. Alzheimer's disease (AD)-like pathology in aged
monkeys after infantile exposure to environmental metal lead
(Pb): evidence for a developmental origin and environmental
link for AD. (2008) J
Neurosci. 2;28(1):3-9.
www.jneurosci.org/cgi/reprint/28/1/3
www.pubmedcentral.nih.gov/picrender.fcgi?artid=2486412&blobtype=PDF
-
Verina,
T, Rohde C A., and Guilarte T R. (2007) Environmental Pb2+
exposure during early life alters granule cell neurogenesis
and morphology in the hippocampus of young adult rats.
Neuroscience. 30; 145(3): 1037–1047. www.pubmedcentral.nih.gov/picrender.fcgi?artid=1892316&blobtype=PDF
-
Ritchie CW, Bush AI, Mackinnon A. Metal-protein
attenuation with iodochlorhydroxyquin (clioquinol) targeting
Abeta amyloid deposition and toxicity in Alzheimer disease:
a pilot phase 2 clinical trial. Arch Neurol
2003;60(12):1685-91. www.medlink.com/cip.asp?UID=14676042
-
Cuajungco
MP, Frederickson CJ, Bush AI. Amyloid-beta metal interaction
and metal chelation. Subcell Biochem 2005;38:235-54. www.medlink.com/cip.asp?UID=15709482
www.ncbi.nlm.nih.gov/sites/entrez
-
Squitti
R, Rossini PM, Cassetta E, et al. d-penicillamine reduces
serum oxidative stress in Alzheimer's disease patients. Eur
J Clin Invest 2002;32:51-9. www.medlink.com/cip.asp?UID=21841062;
www.ncbi.nlm.nih.gov/pubmed/11851727
-
White
LD, Cory-Slechta
DA, Gilbert
ME, Tiffany-Castiglioni
E, Zawia
NH, Virgolini
M, Rossi-George
A, Lasley
SM, Qian
YC, Basha
MR. New
and evolving concepts in the neurotoxicology of lead.
(2007) Toxicol
Appl Pharmacol. 15;225(1):1-27.
PDF
version of this file
|

