|
Environmental
Risks Arising from Changes in Ammunition Materials
Copyright © 2013 –
Cylenchar Limited Dr Peter J Hurley – Cylenchar Limited UK
 European
Soils – Typical Profile and Predicted Impact of Steel Shot
European
Soils – Typical Profile and Predicted Impact of Steel Shot
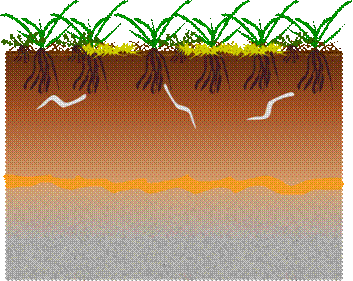
If we consider a
hypothetical ‘typical’ European soil, as we might find on a shooting
range, we usually see: Organic
rich horizons descending into less organic rich layers of weathered soils,
overlying un-weathered substrata and parent rock.
Grasses, herbs, mosses and lichens; rooted in and underlying thatch and
dark organic rich horizon. Surface conditions are typically pH 5.5-7.5. Some
upland areas may be 1 pH unit lower and some Mediterranean coastal fringe
soils a unit higher. We usually find soil invertebrates.
Iron banding from natural weathering is commonly found above less
oxygen rich regions.
 Add steel shot and what is
likely to happen? Steel is not simply an alloy of iron and carbon. Corrosion
will also liberate colloidal oxides of nickel and manganese in addition to
iron. These elements are naturally present in most iron ores and carry
through, or are added to finished steel products.
Accelerated corrosion of legacy lead, and liberation of other heavy
metals naturally present in the soil will occur through redox corrosion and
iron oxide catalysed oxidation. This is an important point as even if you put
pure iron onto the range, such is the power of iron and its oxides as
oxidation catalysts, they will induce breakdown and leaching of previously
stable background metal minerals. In other words: Put a significant amount of
iron on your range and even if you hadn’t a heavy metals leaching problem,
subject to background mineralogy, you are very likely to create one! Add steel shot and what is
likely to happen? Steel is not simply an alloy of iron and carbon. Corrosion
will also liberate colloidal oxides of nickel and manganese in addition to
iron. These elements are naturally present in most iron ores and carry
through, or are added to finished steel products.
Accelerated corrosion of legacy lead, and liberation of other heavy
metals naturally present in the soil will occur through redox corrosion and
iron oxide catalysed oxidation. This is an important point as even if you put
pure iron onto the range, such is the power of iron and its oxides as
oxidation catalysts, they will induce breakdown and leaching of previously
stable background metal minerals. In other words: Put a significant amount of
iron on your range and even if you hadn’t a heavy metals leaching problem,
subject to background mineralogy, you are very likely to create one!
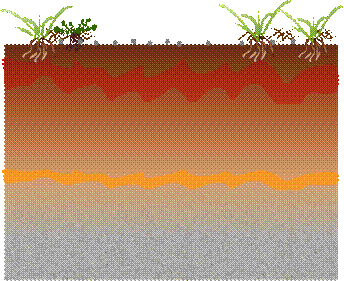 Most
corrosion by-products adhere to colloidal iron oxides, surface clays and
biomass. Initially you might see a reduction in heavy metals leaching.
Iron corrodes to produce alkaline iron (II) compounds which then go on
to acidic iron (III) compounds. Leading to adverse impact on surface soil pH,
swings from 9.5 to 3.5 in days. The overall effect is a long term increase in
site acidity and net increase in metals leaching. We anticipate and observe pH
swings, and the act of corrosion in the soil ‘in and of itself’ takes
oxygen out of the soil raising the level of the soil’s zone of anaerobic
horizon and reducing the zone in which soil invertebrates can thrive.
Iron (II) salts are sold as herbicides, specifically to deter moss. So,
how can you expect steel shot through is corrosion products not to have the
same effect on range soils. We can
anticipate the demise of soil invertebrates due to pH changes and oxidative
stress, with consequential further deterioration in sub-soil drainage.
Reduction of intolerant herbs and deterioration in grass quality and
cover will occur. Formation of
secondary a ‘iron-pan’, leading to deterioration in soil texture (with
iron concretion) will further impede sub-soil drainage, leading to an increase
in surface run-off. The natural consequence of the above is loss of surface
biomass and soils. Most
corrosion by-products adhere to colloidal iron oxides, surface clays and
biomass. Initially you might see a reduction in heavy metals leaching.
Iron corrodes to produce alkaline iron (II) compounds which then go on
to acidic iron (III) compounds. Leading to adverse impact on surface soil pH,
swings from 9.5 to 3.5 in days. The overall effect is a long term increase in
site acidity and net increase in metals leaching. We anticipate and observe pH
swings, and the act of corrosion in the soil ‘in and of itself’ takes
oxygen out of the soil raising the level of the soil’s zone of anaerobic
horizon and reducing the zone in which soil invertebrates can thrive.
Iron (II) salts are sold as herbicides, specifically to deter moss. So,
how can you expect steel shot through is corrosion products not to have the
same effect on range soils. We can
anticipate the demise of soil invertebrates due to pH changes and oxidative
stress, with consequential further deterioration in sub-soil drainage.
Reduction of intolerant herbs and deterioration in grass quality and
cover will occur. Formation of
secondary a ‘iron-pan’, leading to deterioration in soil texture (with
iron concretion) will further impede sub-soil drainage, leading to an increase
in surface run-off. The natural consequence of the above is loss of surface
biomass and soils.
Contents
Environmental Risks Arising from Changes in Ammunition Materials
The Contaminant – Metallic
Lead?
Does Steel Shot Reduce
Environmental Risk?
European
Soils – Typical Profile and Predicted Impact of Steel Shot
Site Risk! - Added Pathways
Mitigation of Site Risk
Copyright © 2013 –
Cylenchar Limited
|

 European
Soils – Typical Profile and Predicted Impact of Steel Shot
European
Soils – Typical Profile and Predicted Impact of Steel Shot Add steel shot and what is
likely to happen? Steel is not simply an alloy of iron and carbon. Corrosion
will also liberate colloidal oxides of nickel and manganese in addition to
iron. These elements are naturally present in most iron ores and carry
through, or are added to finished steel products.
Accelerated corrosion of legacy lead, and liberation of other heavy
metals naturally present in the soil will occur through redox corrosion and
iron oxide catalysed oxidation. This is an important point as even if you put
pure iron onto the range, such is the power of iron and its oxides as
oxidation catalysts, they will induce breakdown and leaching of previously
stable background metal minerals. In other words: Put a significant amount of
iron on your range and even if you hadn’t a heavy metals leaching problem,
subject to background mineralogy, you are very likely to create one!
Add steel shot and what is
likely to happen? Steel is not simply an alloy of iron and carbon. Corrosion
will also liberate colloidal oxides of nickel and manganese in addition to
iron. These elements are naturally present in most iron ores and carry
through, or are added to finished steel products.
Accelerated corrosion of legacy lead, and liberation of other heavy
metals naturally present in the soil will occur through redox corrosion and
iron oxide catalysed oxidation. This is an important point as even if you put
pure iron onto the range, such is the power of iron and its oxides as
oxidation catalysts, they will induce breakdown and leaching of previously
stable background metal minerals. In other words: Put a significant amount of
iron on your range and even if you hadn’t a heavy metals leaching problem,
subject to background mineralogy, you are very likely to create one! Most
corrosion by-products adhere to colloidal iron oxides, surface clays and
biomass. Initially you might see a reduction in heavy metals leaching.
Iron corrodes to produce alkaline iron (II) compounds which then go on
to acidic iron (III) compounds. Leading to adverse impact on surface soil pH,
swings from 9.5 to 3.5 in days. The overall effect is a long term increase in
site acidity and net increase in metals leaching. We anticipate and observe pH
swings, and the act of corrosion in the soil ‘in and of itself’ takes
oxygen out of the soil raising the level of the soil’s zone of anaerobic
horizon and reducing the zone in which soil invertebrates can thrive.
Iron (II) salts are sold as herbicides, specifically to deter moss. So,
how can you expect steel shot through is corrosion products not to have the
same effect on range soils. We can
anticipate the demise of soil invertebrates due to pH changes and oxidative
stress, with consequential further deterioration in sub-soil drainage.
Reduction of intolerant herbs and deterioration in grass quality and
cover will occur. Formation of
secondary a ‘iron-pan’, leading to deterioration in soil texture (with
iron concretion) will further impede sub-soil drainage, leading to an increase
in surface run-off. The natural consequence of the above is loss of surface
biomass and soils.
Most
corrosion by-products adhere to colloidal iron oxides, surface clays and
biomass. Initially you might see a reduction in heavy metals leaching.
Iron corrodes to produce alkaline iron (II) compounds which then go on
to acidic iron (III) compounds. Leading to adverse impact on surface soil pH,
swings from 9.5 to 3.5 in days. The overall effect is a long term increase in
site acidity and net increase in metals leaching. We anticipate and observe pH
swings, and the act of corrosion in the soil ‘in and of itself’ takes
oxygen out of the soil raising the level of the soil’s zone of anaerobic
horizon and reducing the zone in which soil invertebrates can thrive.
Iron (II) salts are sold as herbicides, specifically to deter moss. So,
how can you expect steel shot through is corrosion products not to have the
same effect on range soils. We can
anticipate the demise of soil invertebrates due to pH changes and oxidative
stress, with consequential further deterioration in sub-soil drainage.
Reduction of intolerant herbs and deterioration in grass quality and
cover will occur. Formation of
secondary a ‘iron-pan’, leading to deterioration in soil texture (with
iron concretion) will further impede sub-soil drainage, leading to an increase
in surface run-off. The natural consequence of the above is loss of surface
biomass and soils.